This story was originally published on MedTech Dive. To receive daily news and insights, subscribe to our free daily MedTech Dive newsletter.
Dive Brief:
-
The company plans to submit an investigational device exemption to the Food and Drug Administration. If approved, Glucotrack expects to begin the study in the second half of the year.
-
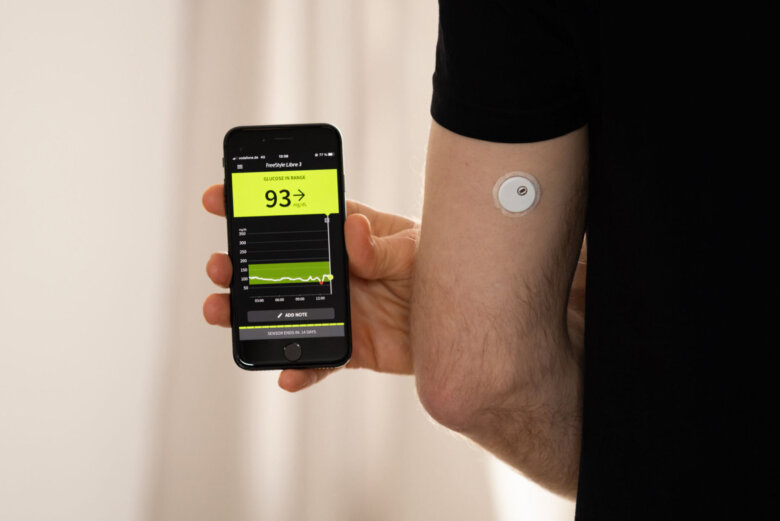
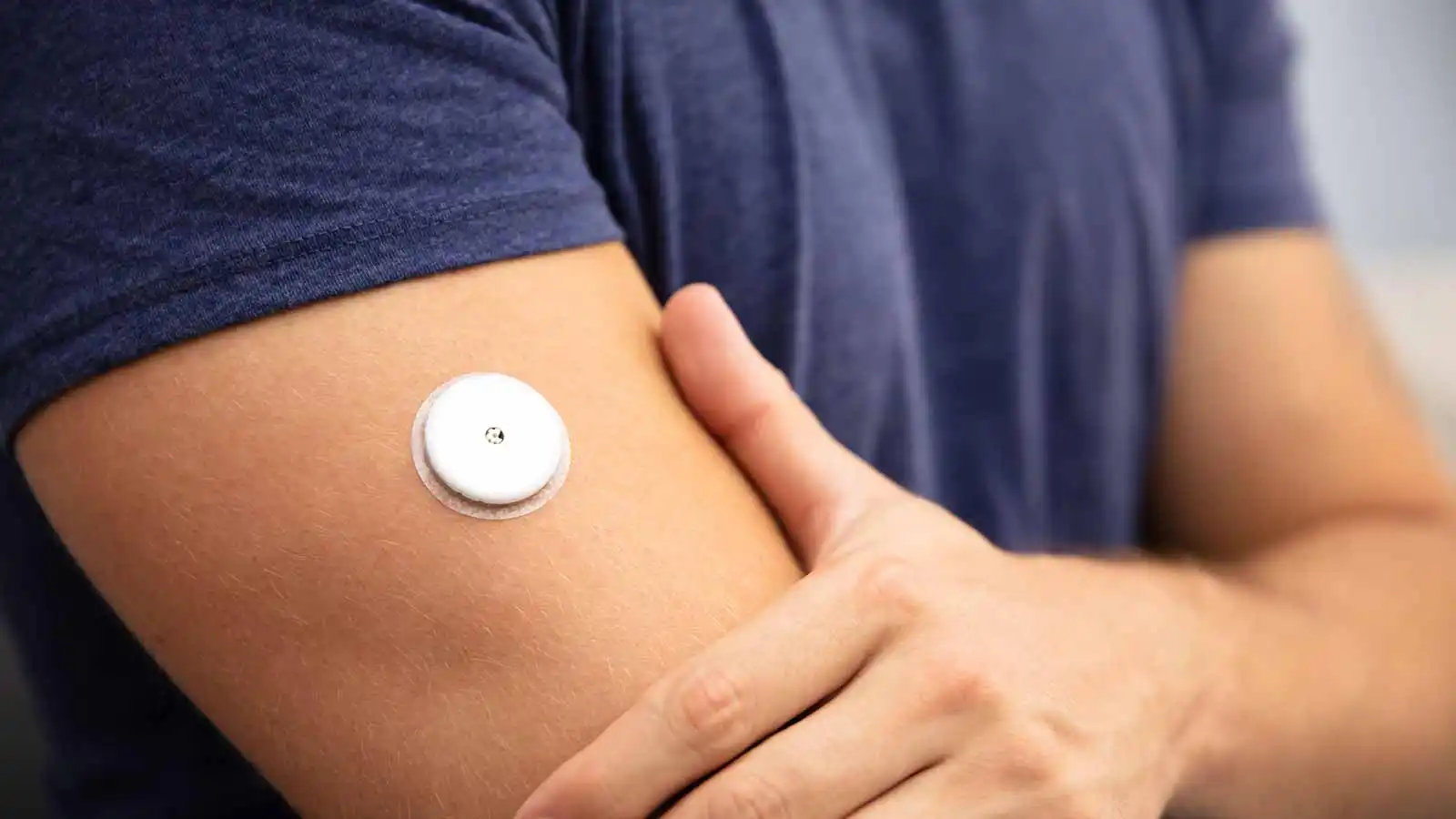
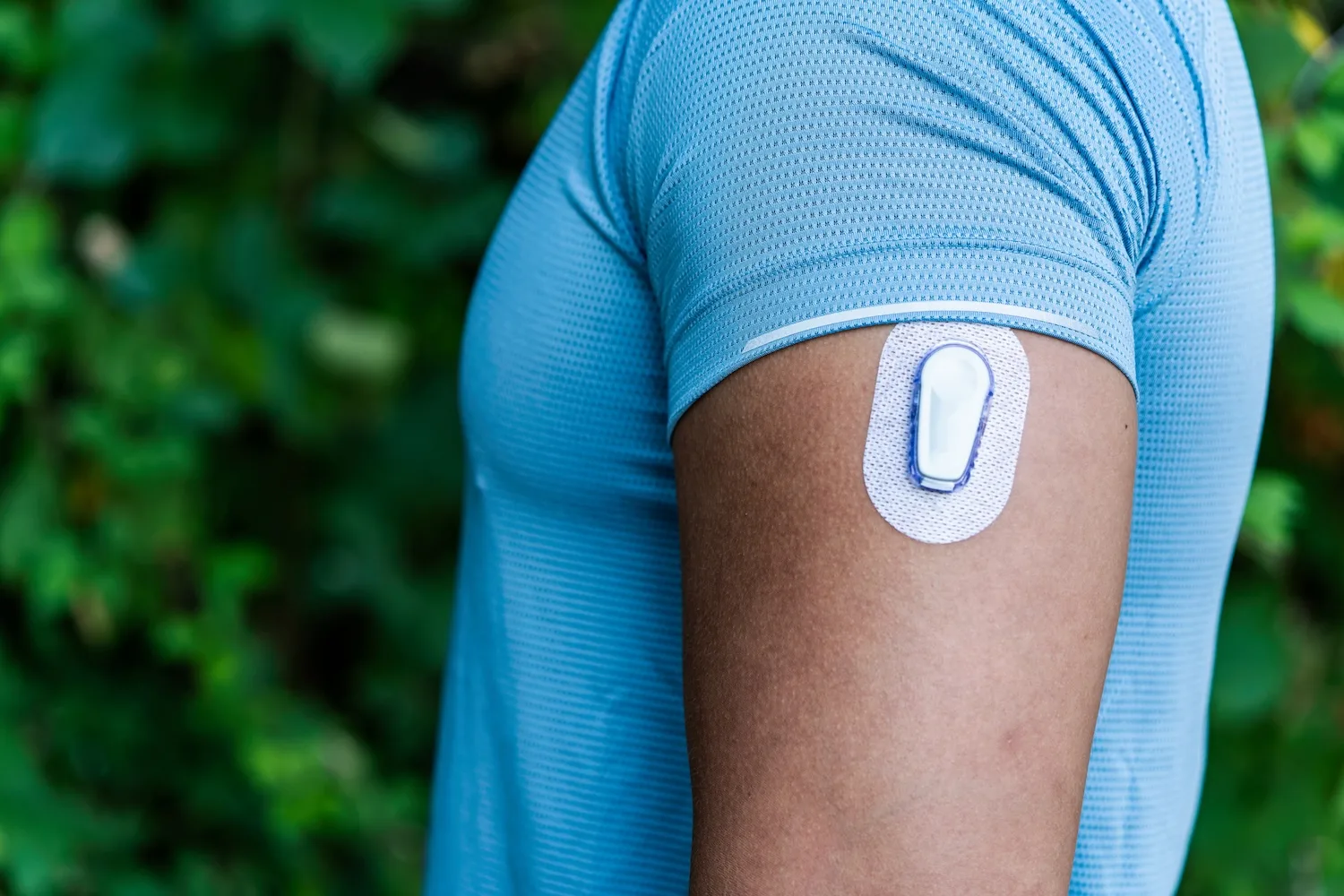
Glucotrack’s device differs from other continuous glucose monitors on the market by measuring glucose values directly from the blood, rather than the interstitial fluid under the skin. The device is implanted in the subclavian vein near the collarbone, and is intended to last for three years.
Dive Insight:
Glucotrack said it is ready to start a U.S. clinical trial following progress in other countries. Last year, the company shared results of a first-in-human trial of the device in Brazil.
Advertisement
Advertisement
During the five-day trial, the 10 participants had no procedure- or device-related adverse events, and the device demonstrated a mean absolute relative difference, or MARD, of 7.7%. MARD is an accuracy measurement for CGMs showing the difference between CGM readings and reference blood glucose values.
The company started another feasibility study in Australia in July, with similar performance to the trial in Brazil.
For the planned U.S. study, Glucotrack said it has secured a clinical trial site and found a contract research organization experienced with diabetes technology. The company has also worked on the design of its CGM to improve its functionality and performance.
“With trial infrastructure in place and product enhancements completed, we are prepared to initiate the study, pending FDA approval,” CEO Paul Goode said in a statement.
Recommended Reading